10 Interesting Facts about the Navy
Facts about The Navy tell you about the USN or United States Navy. It is included as one of the 7 uniformed services in US. It is one of the branches of United Armed Forces. The US Navy is famous in the world. Do you know that it has the largest aircraft carrier fleet in the world? There are ten aircraft carrier fleets in service, while the two are under the construction. Here are other interesting facts about the navy for you: Facts about The Navy Read More ... »
10 Interesting Facts about the Murray Darling Basin
Let me show you the rivers in Australia in Facts about The Murray Darling Basin. The name of the basin is taken from the name of the two famous rivers in Australia. Both are the Murray River and Darling River. They are located in the interior of southeastern Australia. Here are other facts about Murray Darling basin below: Facts about The Murray Darling Basin 1: the land mass The basin is very important to the life of the Australian people. Do you Read More ... »
10 Interesting Facts about the Murray River
Facts about The Murray River present the detail about the longest river in Australia. It has the length at 1,558 miles or 2,508 kilometers. This river creates a border between Victoria and New South Wales. There are several lakes which get the water from the flow of Murray River such as Lake Alexandrina and The Coorong. Find out more facts about Murray River below: Facts about The Murray River 1: the irrigated region The icon of food bowl of Australia Read More ... »
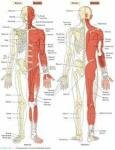
10 Interesting Facts about the Musculoskeletal System
Facts about The Musculoskeletal System talk about the system of organ which gives the human being the ability to move both the skeletal and muscular systems. You can also call it as the locomotor system. In the past, people often called it the activity system. Get more facts about musculoskeletal system below: Facts about The Musculoskeletal System 1: the function Let’s find out the function of the musculoskeletal system first. The human being can Read More ... »
10 Interesting Facts about the Muscles
Facts about The Muscles give you the interesting facts about the parts of human body. Inside the human body, we have at least 600 muscles. The function of the muscles is various. It can be used to help us lifting the heavy object, walk, and run, pump the blood, keep the heart beating and move the food to the intestines. Here are some facts about muscles for you: Facts about The Muscles 1: the controlled and uncontrolled muscles Some muscles in the Read More ... »
10 Interesting Facts about the Mughal Empire
Facts about The Mughal Empire talk about the most powerful Islamic state which ruled India. It was established in 1526. During the 17th century, it reached the pinnacle. The famous building of Mughal Empire is Taj Mahal. Have you ever visited this building? It is one of the famous landmarks in India today. Many people love to visit Taj Mahal. Facts about The Mughal Empire 1: the meaning of Mughal Mughal has the meaning Mongol. The Mongol warlords Read More ... »
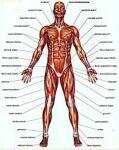
10 Interesting Facts about the Muscular System
Facts about The Muscular System show you that muscles are very important for human being. When they want to do the physical movement, the muscles should work. It can keep our heart beat normally. If there is something wrong with the muscle, you have to be careful. Let me show you some interesting muscular system facts below: Facts about The Muscular System 1: definition of muscles What is muscle? Actually it is a soft tissue that you can find Read More ... »
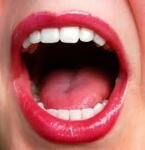
10 Interesting Facts about the Mouth
Facts about The Mouth tell you about a part of human face that you can use to smile, frown, talk, or even whistle. The mouth has to work with the tongue and teeth if we want to eat food or even speak. This part of the body is very important. It will be difficult for us to eat and speak if the mouth is not functioned well. Here are facts about mouth for you: Facts about The Mouth 1: chewing food When we chew food, the mouth should work together with Read More ... »
8 Interesting Facts about the Moon Landing
Let me show you one of the interesting histories of the mankind in Facts about The Moon Landing. The moon landing gains a lot of attention from the people in the world after Neil Armstrong walked on the moon for the first time. There were more than 600 million people in the world watched the moon landing. Find out more facts about Moon landing below: Facts about The Moon Landing 1: the first moon landing The first manned moon landing was conducted Read More ... »
8 Interesting Facts about the Moose
Facts about The Moose elaborate the details about one of the members in deer family. You can find it living in the colder climate area in Northern hemisphere. When the summer season comes, the moose can be seen in living near the streams and rivers. When the winter comes, they live in the forest. Let’s find out more facts about moose below: Facts about The Moose 1: the height of male and female moose The male and female moose have the similar height Read More ... »







 www.PortlandPayday.Loans
www.PortlandPayday.Loans